How Low Back Pain is Affecting Millions of Americans?

Back pain
is the leading cause
of missing work.

200 million days
in bed a year.
Adults with back pain spend almost

16 million Americans
have chronic back pain.

200 million
days in bed
a year.
sadness, worthlessness, and hopelessness that interfere with life.
You Want to Help?
Knowledge is power.
By participating in the all on-line BACKHOME study, you will contribute to our knowledge about low back pain, which will help our research team develop better treatments for you and others like you.

About the online study
You are eligible for BACKHOME if you:
- Are 18 years or older
- Have had low back pain for three months or more
- Live in the United States
Don't let chronic low back pain hold back present and future generations!
By taking part in BACKHOME, you will contribute to research that will help millions of people who, like you, suffer from chronic low back pain.
We’re in it together to find better ways of helping people who have chronic low back pain!

Complete the online surveys and help researchers
The study will be done completely online and there will be no in-person visits.

Learn about new treatments and access digital tools
Our team will keep you updated about the latest news and discoveries in the field of back pain research.

Be the first invitees for future studies
BACKHOME participants will be invited to new clinical trials and studies in the future.
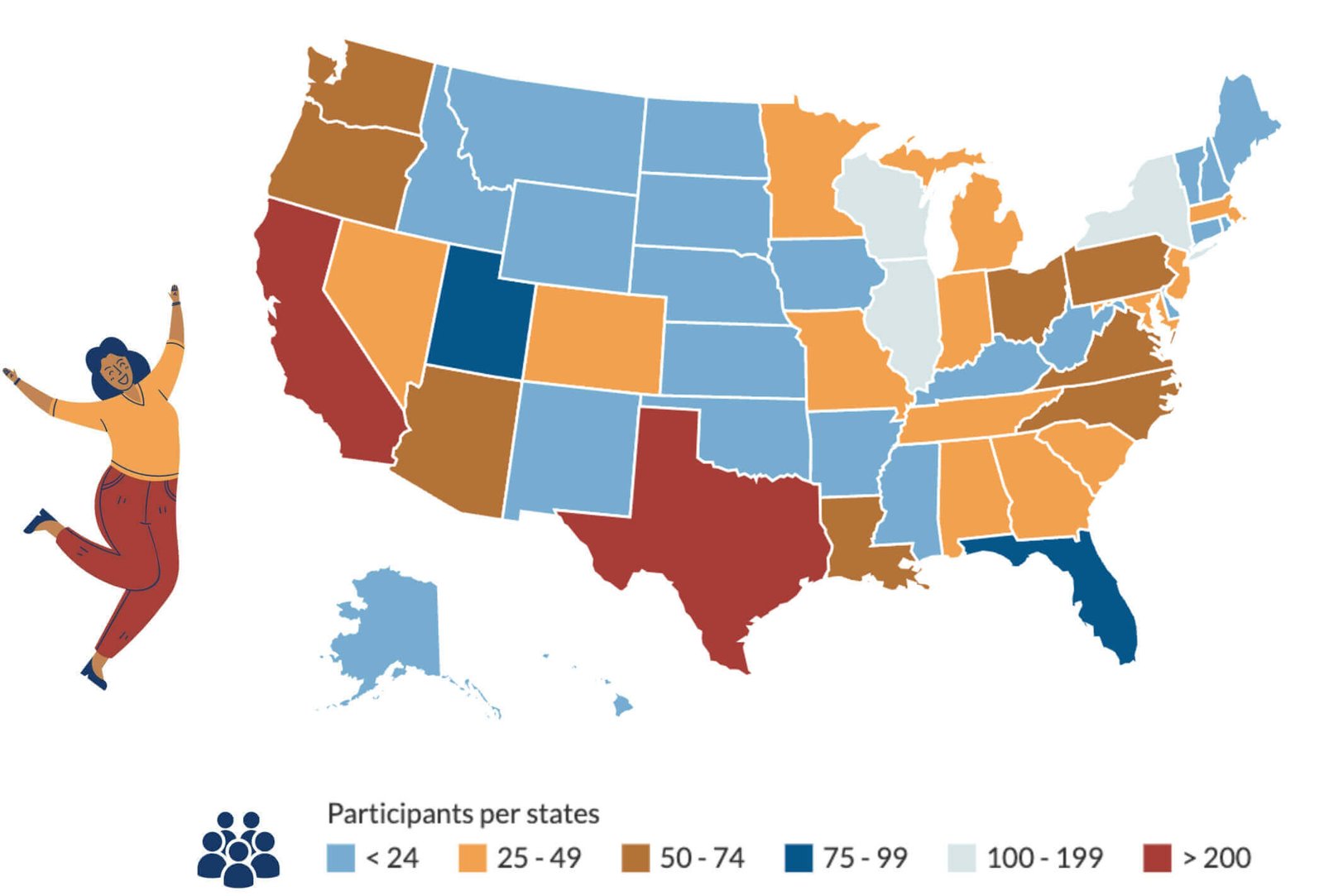
Thank you to all the BACKHOME participants who have joined from across the US.
We now have BACKHOME participants in every state in America.

A Word From Our Team
A Message from the Patient Advisory Board
The Research Team
Frequently Asked Questions
How do I register for the study?
It’s easy to register. Click here to Enroll Now. This will take you to our secure Eureka research platform. You will be asked to provide your name, email (a simple way for us to contact you), birth date, and a password. You’ll get a confirmation email to verify your email address, then you will complete the electronic consent and can start taking the study surveys.
Why do I have to sign a consent form?
Once you register, you’ll be taken to the consent page. This consent page provides you with an outline of the study and details exactly what your role will be. It is important that you have this information to make an informed decision about participating.
What do I need to do if I participate in the study?
If you join the BACKHOME study, you’ll be asked to complete an initial online study visit, which includes several short easy-to-complete surveys. We will then contact you by email, every 3-6 months, with health update surveys. You may also receive special invitations to participate additional parts of the study. Some examples of future project include clinical trials and studies of mobile apps to track your sleep, sensors to measure activity, DNA studies, and digital back pain programs.
What’s expected of me?
We’ll contact you by email (or via the Eureka mobile app, if applicable) every 3-6 months over the next two years. We hope you will be able to answer the health-related surveys and if you receive special invites from us to join additional parts of this study, we hope you will be interested! All of these activities will help us track how an individual’s back pain evolves over time and what treatments you may be using as a part of your normal health care.
What are the surveys all about?
You will complete all of the surveys online (or on your smartphone) at your leisure. Most of the surveys are very basic and can be done in 5 minutes. Some of them may ask for sensitive information, such as regarding your mood and your sleep. All of your data will be kept secure.
How often do I have to take these surveys?
You will be contacted to complete an eVisit about every 3-6 months. The first survey will take the most time to complete because we’d like to learn about you and what your back pain experience is. The subsequent surveys will be much shorter in length. You can fill out the surveys in each eVisit all at once or spread them out over a few days. In addition, you’ll receive occasional special invitations to participate in sub-studies or take special surveys, depending on your specific conditions.
Is my data safe?
Absolutely. We take this VERY seriously and do everything we can to protect your information and keep it private and secure. The Eureka platform uses a state-of-the-art HIPAA compliant security system and all sensitive data is encrypted.
How will my information be kept confidential?
If information from this study is published or presented at scientific meetings, your name and other personal information will never be used or released. This research is covered by a Certificate of Confidentiality from the National Institutes of Health. This means that the researchers cannot release or use information that may identify you in any action or suit unless you say it is okay. They also cannot provide them as evidence unless you have agreed. This protection includes federal, state, or local civil, criminal, administrative, legislative, or other proceedings.
What is my information used for?
We will only use your information for research purposes. Your survey responses will be used in grouped research analyses, and the results of these analyses may be presented at scientific conferences, published, and used to help develop new treatments to help improve the care of patients with back pain like you. These presentations and publications will never include any information that could identify you.
What about my privacy?
Protection Against Involuntary Disclosure of Your Information: To help us protect your privacy, we have applied for a Certificate of Confidentiality from the United States Department of Health and Human Services (DHHS). This certificate protects against compulsory legal demands, such as court orders and subpoenas, for identifying information or identifying characteristics of a research participant. The BACKHOME Study staff can’t be forced to disclose study information that identifies you, even if ordered by a court subpoena, in any federal, state, or local civil, criminal, administrative, legislative, or other proceedings. The researchers will use the certificate to resist any demands for information that would identify you, except to prevent serious harm to yourself or others. We will, in all cases, take the necessary action, including reporting to authorities, to prevent serious harm to yourself or others.
Do you follow HIPAA rules?
Electronic Security and Adherence to the HIPAA Privacy Rule: The BACKHOME study follows the security guidelines of the U.S. Health Insurance Portability and Accountability Act of 1996 (HIPAA). All study data is transmitted, stored, and processed in a secure environment.
What if I have questions about the study?
If you have questions about the study before you sign up, or if you have difficulty signing up, please use the form below to contact us. Or you can also email us at BackHomeHelpDesk@ucsf.edu or call (U.S. toll free) 1-844-224-6634.













